-
E-mail
chenfeichen@huaduanbio.com
-
Phone
18805736021
-
Address
Room K16, 4th Floor, Building 2, No. 4 Jinjiadu South Road, Liangzhu Street, Yuhang District, Hangzhou City, Zhejiang Province
Hangzhou Huaduan Biotechnology Co., Ltd
Fully automatic pathogen immune rapid screening device
- Model
- Nature of the Manufacturer
- Producers
- Product Category
- Place of Origin
HD-IMseriesFully automatic pathogen immune rapid screening device
1. Main purpose:Pathogenic bacteria and toxins in foodfastdetectionAnd in the field of immunoassays
2. Standard certification:HD-IMseriesFully automatic pathogen immune rapid screening devicepassNational StandardGB/TAuthentication method,AOACTheFDAandISOStandard Certification
3. Technical Indicators:
3.1 hardware performance
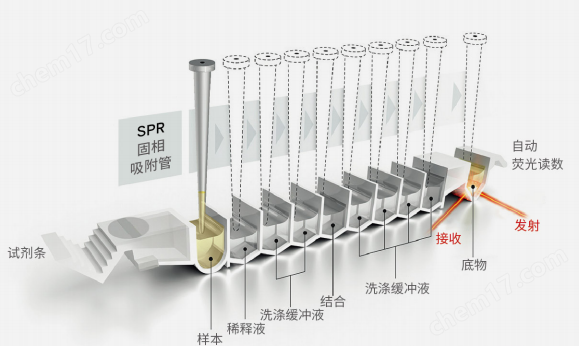
3.1.1Automation: The ready to use reagent strip is inserted into the instrument, and the system automatically performs different steps such as preheating, sample aspiration, measurement, analysis, and printing results for the entire immunoassay.
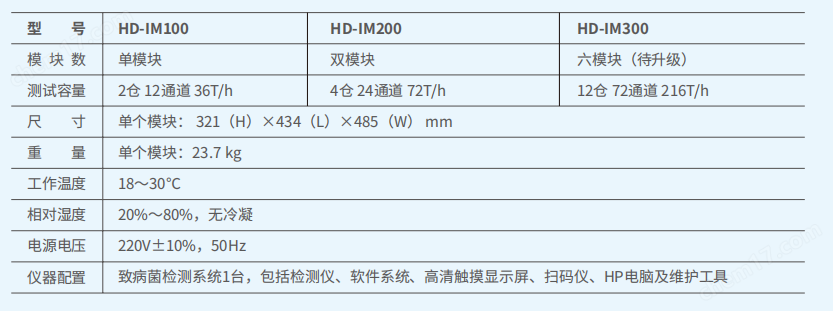
3.1.2Test capacity: Modular design, flexible test capacity, can be conducted simultaneously12A different testStackable design saves space,Support subsequent upgrade and expansion.
3.1.3Sample absorption system: The instrument automatically performs the processes of sample mixing, dilution, cleaning, and transfer in immunoassay;Intelligent pump function, automatic blockage inspection to avoid cross contamination.
3.1.4Instrument StatusLEDThe instrument is equipped with multipleLEDUsed to indicate the overall status of the instrument and the operation status of the storage compartment, and automatically warn of instrument blockage and leakage.
3.1.5Reaction zone: The system is a multi parameter analyzer, with each module having2An independent reaction chamber, each containing6Each experimental channel operates independently and temperature controls independently, can be started separately, and can carry out the same or different testing projects simultaneously or at any time.
3.1.6The central system of the instrument controls the temperature of the built-in incubator35.3℃to38.7℃ ± 0.7℃
3.1.7Detection system: A fluorescence scanner is installed inside the instrument to detect changes in fluorescence at the end of each reagent strip. Maximum detection per hour36Next, the result report time is from30Minutes to arrive2hour, 120test/sky/module
3.1.8Sleep mode: One or more modules in the instrument group can be selected to automatically enter sleep mode when not in use, without affecting the normal operation of other modules, saving energy, and effectively shortening the preheating time for instrument startup.
3.1.9Internal camera control is used to automatically identify the main consumables in the reaction area, avoiding mixed placement of different batch numbers and testing items due to misoperation
3.2 Software features
3.2.1Includes high-definition touch display and internal computer for individual modules, performing interface functions between users and instruments, detecting reaction processes, data management, storing and printing results, as well asLIMSCommunicate.
3.2.2Can store pre-set standard curves, each14sky-28Only one single point correction is needed for the day. Cross module sharing of calibration dataNo need for repeated calibration.
3.2.3Support bidirectional connection of laboratory data management system.
3.2.4Provide compliance21CFR 11The required functionality meets the audit tracking requirements and ensures data integrity.
3.3 detection performance
3.3.1Reagent strip: It is packaged in a closed and independent single serving container, and each kit should be equipped with both calibration and quality control solutions. There is no need to purchase any other test consumables such as colorimetric cups or pipettes separately.
3.3.2Suitable for various environments and food substrates, throughISOTheAOACWaiting together40Multiple international certifications,24Hours and48Hourly solutions are available for selection.
3.3.3Can cause hepatitis、AIDSSerological testing, bacterial and viral antigen testing, fertility testing/Pregnancy examination, thyroid hormones, allergic reactions, tumor markers and other immunological tests.
4.3.3various, multiple, diverse, a variety ofTest strip categories are available for selection, including
★Salmonella detection (antibody technology)/Clone phage binding protein)
★Listeria testing/Detection of Listeria monocytogenes
★Escherichia coliO157Type detection (antibody technology)/Clone phage binding protein)
★Staphylococcus enterotoxin detection
★ Toxoplasma gondiiIgMantibody test
★ Rubella virusIgMantibody test
★ Alpha fetoprotein testing
★ Detection of carcinoembryonic antigen
4. Technical data and specifications: