-
E-mail
hanswu1225@163.com
-
Phone
13916002376
-
Address
Room 302, Building B, Yanke Plaza, No. 1111 Xinzhen Street, Xinqiao Town, Songjiang District, Shanghai
Shanghai Kerui Instrument Technology Co., Ltd
Abbemat Professional 7001 Pharmaceutical Refractometer
- Model
- Nature of the Manufacturer
- Producers
- Product Category
- Place of Origin
Abbemat Professional 7001 Pharmaceutical Refractometer
Capable of meeting pharmaceutical industry regulations and other environmental regulatory requirements: 21 CFR Part 11, GAMP 5, and EU GMP Vol. 4 Annex 11. They can provide secure data management, audit trails, electronic signatures, and access control to ensure audit readiness at all times, and can be integrated with processes to securely store measurement values and metadata. The AISQ+certification package increases certification speed by 70% and provides risk analysis, SOP, and traceability documents for rapid deployment in regulatory environments
Abbemat Professional 7001 Pharmaceutical Refractometer
 Fully certify your pharmaceutical productsrefractometer
Fully certify your pharmaceutical productsrefractometer
The performance evaluation and daily monitoring of measuring equipment is not just about verifying refractive index. Anton Paar also ensures the accuracy of the second key parameter: temperature.
Anton Paar service engineers can use the T-Check system to verify accurate temperature measurements, providing:
On site traceable temperature verification and calibration
High accuracy prism surface temperature measurement
Prevent measurement errors related to temperature
Complete documents and authentication
 Prevent errors through user guides
Prevent errors through user guides
User permission management ensures that operators can only access the functions necessary for their work. Only users with corresponding permissions can change settings. User profiles can be seamlessly integrated with domains.
Conventional measurements can be constructed based on your SOP and pre-defined with qualified/unqualified standards. You can configure the inspection interval to perform system suitability testing based on your measurement device control protocol.
 Security Data Management Options
Security Data Management Options
Automatic refractometers can be used independently and export file based data to your network. Alternatively, data can also be managed through AP Connect software: an encrypted SQL database that is not easily manipulated ensures secure storage of data throughout the retention period, direct backup and recovery of data, and easy access to data.
 Electronic AISQ+verification file
Electronic AISQ+verification file
comprehensiveAnalytical instrument and system validation(AISQ+) greatly reduces the workload required for using newly manufactured pharmaceutical refractometers. It can be printed orallDigitize electronic signatures to accelerate the review and approval process during qualification examination.
 Durable sapphire prism - scratch resistant, 25 year warranty
Durable sapphire prism - scratch resistant, 25 year warranty
The high-quality optical components of the automatic pharmaceutical refractometer are carefully designed and durable, ensuring excellent elasticity and long service life.
Cleaning can be done using a scraper and other laboratory tools
The Mohs hardness is nine, close to diamond level
Prism comes with a 25 year warranty
Specifications
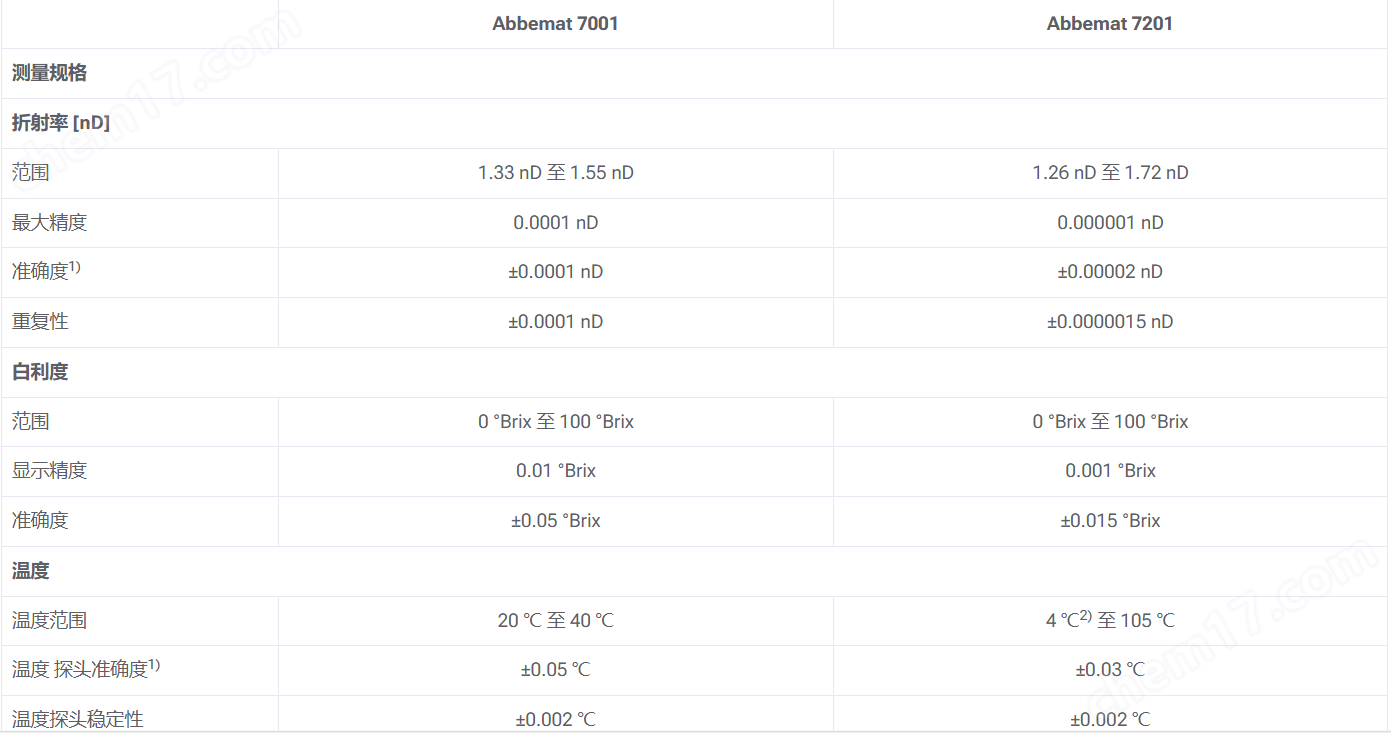


1) Effective under standard measurement conditions (T=20 ℃, ambient temperature between 20 ℃ and 25 ℃)
2) The maximum ambient temperature is 35 ℃






